Amaryl 4mg 30 Tablets
5 Produkte wurden in den letzten 18 Stunden verkauft
Verkauft sich schnell! Über 5 Personen haben in ihrem Warenkorb
28 Personen sehen sich das gerade an
- Estimated Delivery : Same day
- Free Shipping & Returns : On all orders over 200 EGP










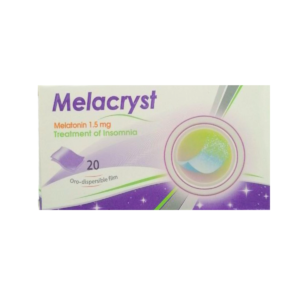
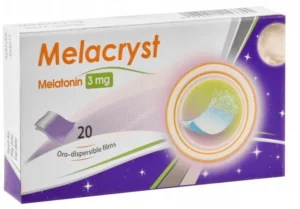


Bewertungen
Es gibt noch keine Bewertungen.