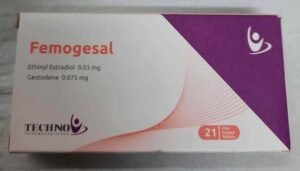
Femogesal 21 Tablets
13 products sold in last 5 hours
Selling fast! Over 20 people have in their cart
30 Personen sehen sich das gerade an
- Estimated Delivery : Same day
- Free Shipping & Returns : On all orders over 200 EGP







Bewertungen
Es gibt noch keine Bewertungen.